11-year-old female spayed Poodle presented for onset of seizure activity
History
An 11-year-old female spayed Poodle presented for an onset of seizure activity over the previous 2 months with a post-ictal period of approximately one hour. The dog had been initiated on twice daily oral phenobarbitone at the onset of the seizures although seizure frequency was increasing. The owner was also concerned about the possibility of the dog having neck pain although a neurological examination was considered normal at the time of presentation. Otherwise considered healthy, the dog underwent a brain MRI using the Hallmarq 1.5T machine (see Figs 1 & 2).
The imaging results
Figure 1. (A) Transverse T2W, (B) Transverse FLAIR, (C) Transverse T1W pre-contrast and (D) Transverse T1W post-contrast images of the brain at the level of the frontal lobe. Red arrow – perilesional oedema; Blue arrow – mass lesion; Yellow arrow – midline shift.
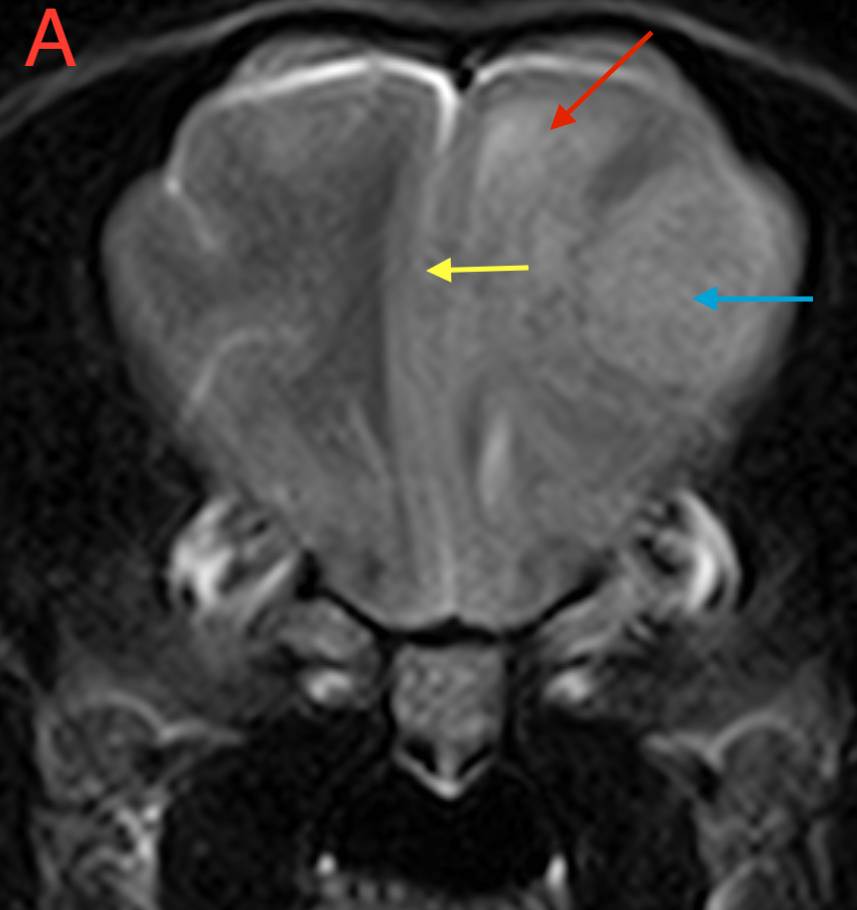
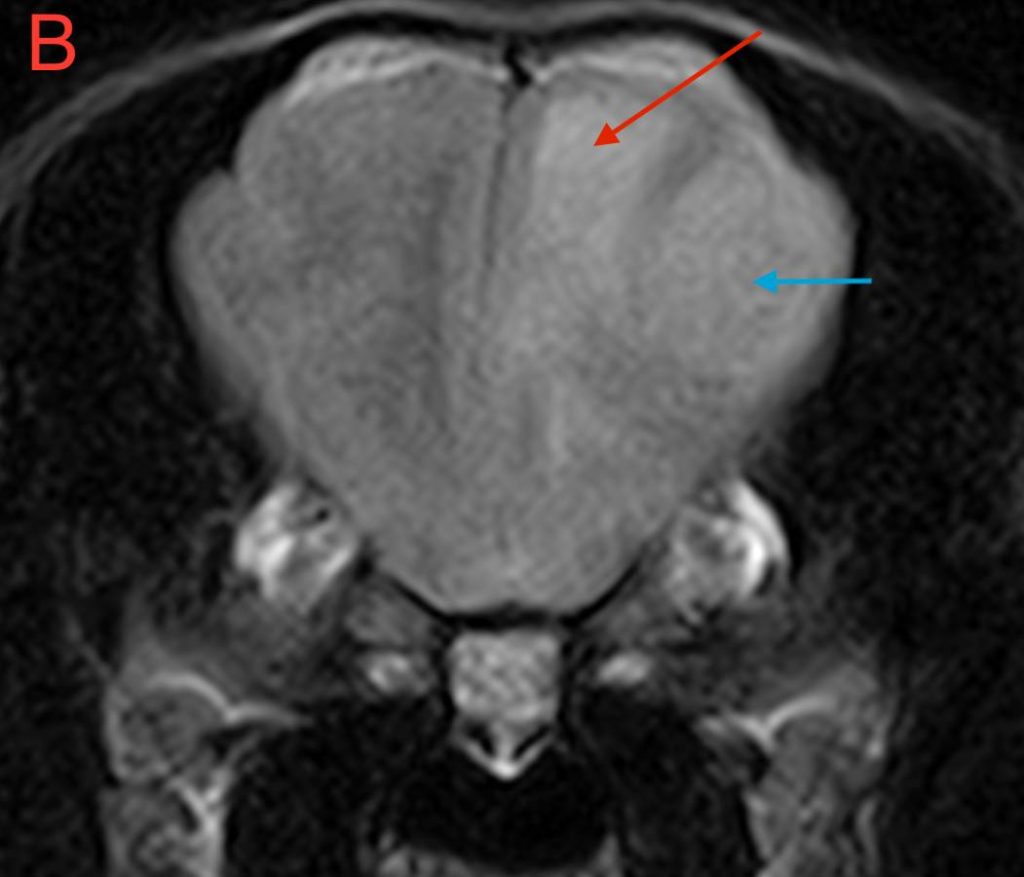
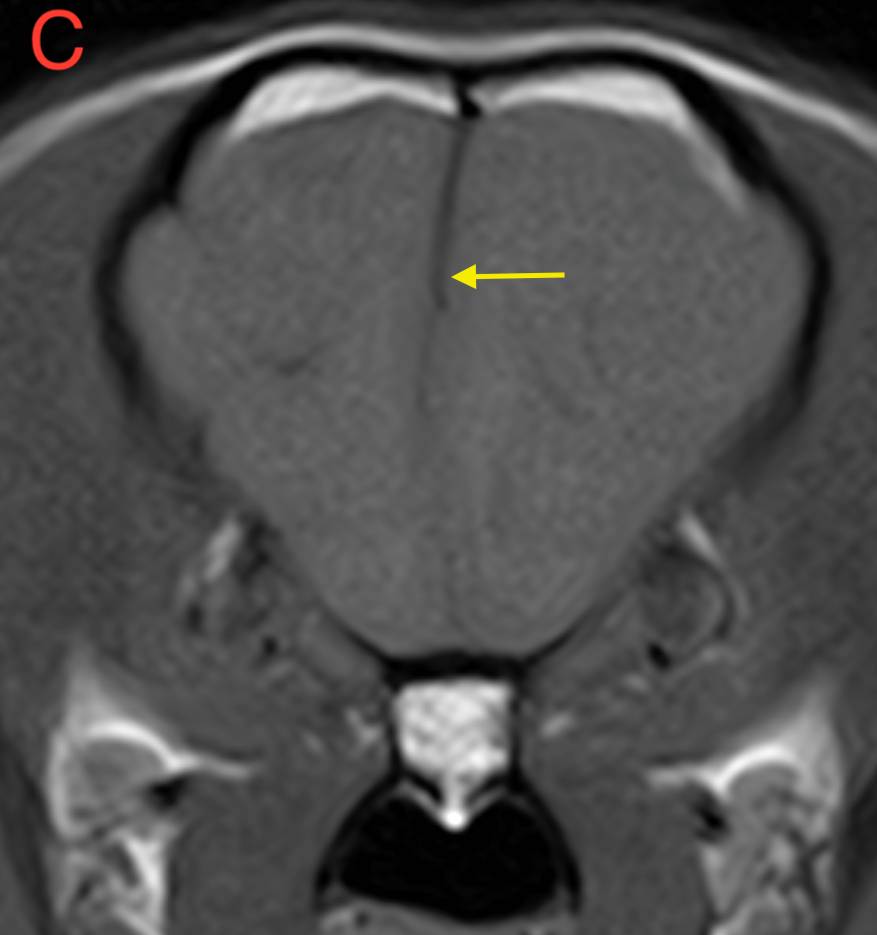
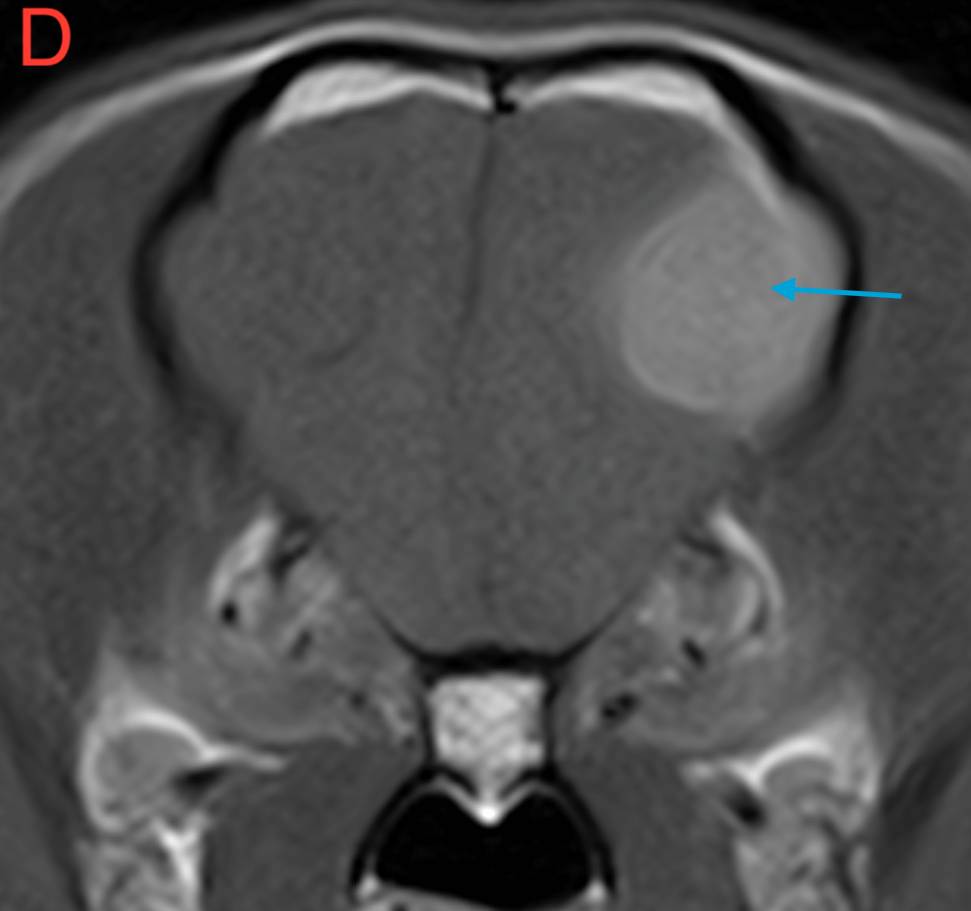
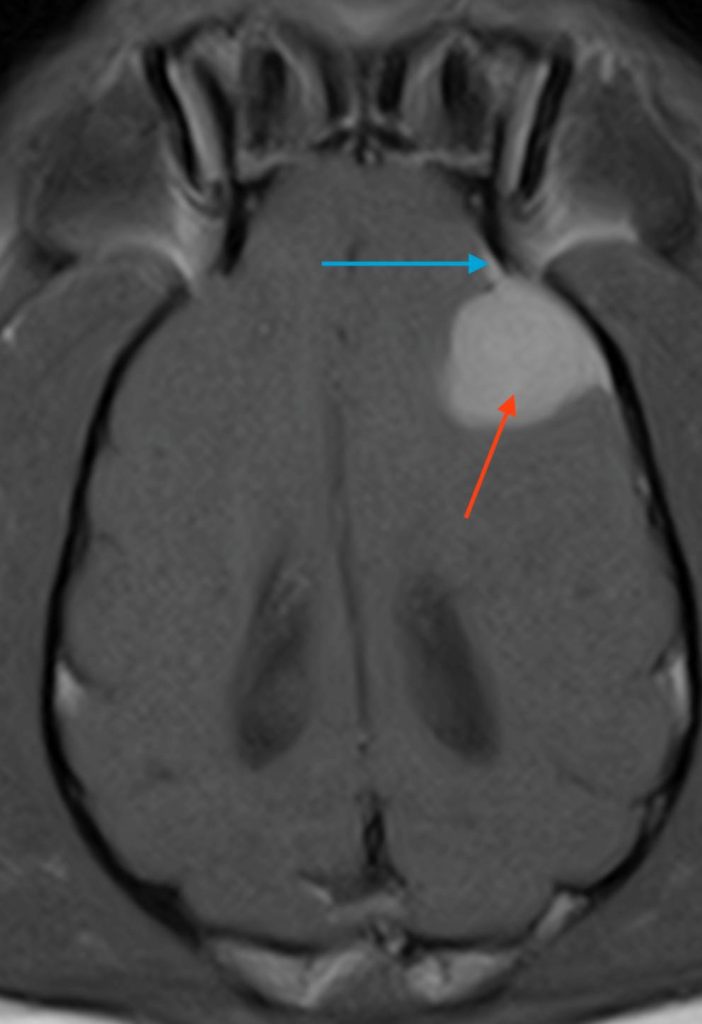
Figure 2. Dorsal T1W images of the brain (A) pre-contrast and (B) post-contrast. Blue arrow – dural tail; Red arrow – mass lesion.
There is an extra-axial and broad-based space-occupying lesion, centered on the left frontal lobe. This lesion is well-defined and it shows a rounded to oval shape. It is homogeneous, hyperintense in T2W when compared to grey matter, hyperintense in FLAIR and isointense in T1W. After contrast, there is moderate to severe and homogeneous contrast enhancement. Caudal and rostrally, a short dural tail sign is seen, suggesting attachment to the meninges.
This lesion causes moderate perilesional oedema which tracks along the white matter to reach the rostral occipital lobe, and causes mild mass effect, with mild midline shift. There is no evidence of transtentorial or cerebellar herniation. No other intracranial or extracranial lesions are seen.
The main differentials for this mass lesion include meningioma, histiocytic sarcoma, granular cell tumor and lymphoma, with meningioma being the most common possibility.
The most valuable MRI sequence for this block
The T1W post-contrast sequence demonstrates both meningeal involvement and the homogenous enhancement often seen associated with tumours such as meningioma originating outside of the blood-brain barrier. Typically, the extent of disease can be accurately assessed as the tissue which enhances. The extension of the mass lesion along the meninges as a ‘dural tail’ can be seen clearly in Fig 2B and is often a characteristic of meningiomas, although not 100% specific for this tumour type.
Disease overview
Meningiomas are the most common brain tumours in dogs and cats and typically affect older animals (>10 years of age). Golden retrievers, boxers, miniature schnauzers, and rat terriers have been identified as breeds in which intracranial meningiomas are overrepresented.
These tumours arise from the arachnoid layer of the meninges, originating at the periphery of the brain parenchyma and expanding inwards from their extra-axial location. Most canine meningiomas are adjacent to the calvarium and a significant number of these tumours involve the olfactory/frontal region, the floor of the cranial cavity, the optic chiasm or the suprasellar and parasellar regions. In cats, common locations include the third ventricle, the rostrotentorial meninges and, rarely, the cerebellar meninges. In dogs, meningiomas have a tendency to infiltrate into the brain tissue but in cats, meningiomas are often well encapsulated.
Typically on MR imaging, meningiomas are solitary, broad-based, well-marginated, extra-axial contrast-enhancing masses. These tumours may contain intratumoural fluid/cysts, mineralization, haemorrhage, or be predominantly cystic. Meningiomas may also be associated with calvarial hyperostosis or a dural tail sign. Although the dural tail feature is commonly associated with meningiomas, it is not specific for this tumour type, or for neoplastic diseases in general. Peritumoural oedema is present in >90% of canine meningiomas and is extensive and diffuse in many cases.
Studies that have included cohorts of dogs with rostrotentorial meningiomas provide median survival times ranging from 7-14 months when surgery is used as a sole modality and there is some evidence that improving surgical excision using ultrasonic aspiration or endoscopy may result in prolonged median survival times. Radiation therapy alone has resulted in average survival times of over 17 months for dogs with intracranial meningiomas.
With thanks to the team at Dogwood Referrals, Manchester, UK for providing this case study. All images seen here were taken with a Hallmarq Small Animal 1.5T MRI machine.
References
- Miller AD, Miller CR, Rossmeisl JH. Canine Primary Intracranial Cancer: A Clinicopathologic and Comparative Review of Glioma, Meningioma, and Choroid Plexus Tumors. Front Oncol. 2019 Nov 8;9:1151
- Motta L, Mandara MT, Skerritt GC. Canine and feline intracranial meningiomas: an updated review. Vet J. 2012 May;192(2):153-65.